 
The viscosity of water decreases smoothly from the freezing point while the density remains essentially constant except for the small maximum in density near 4☌. 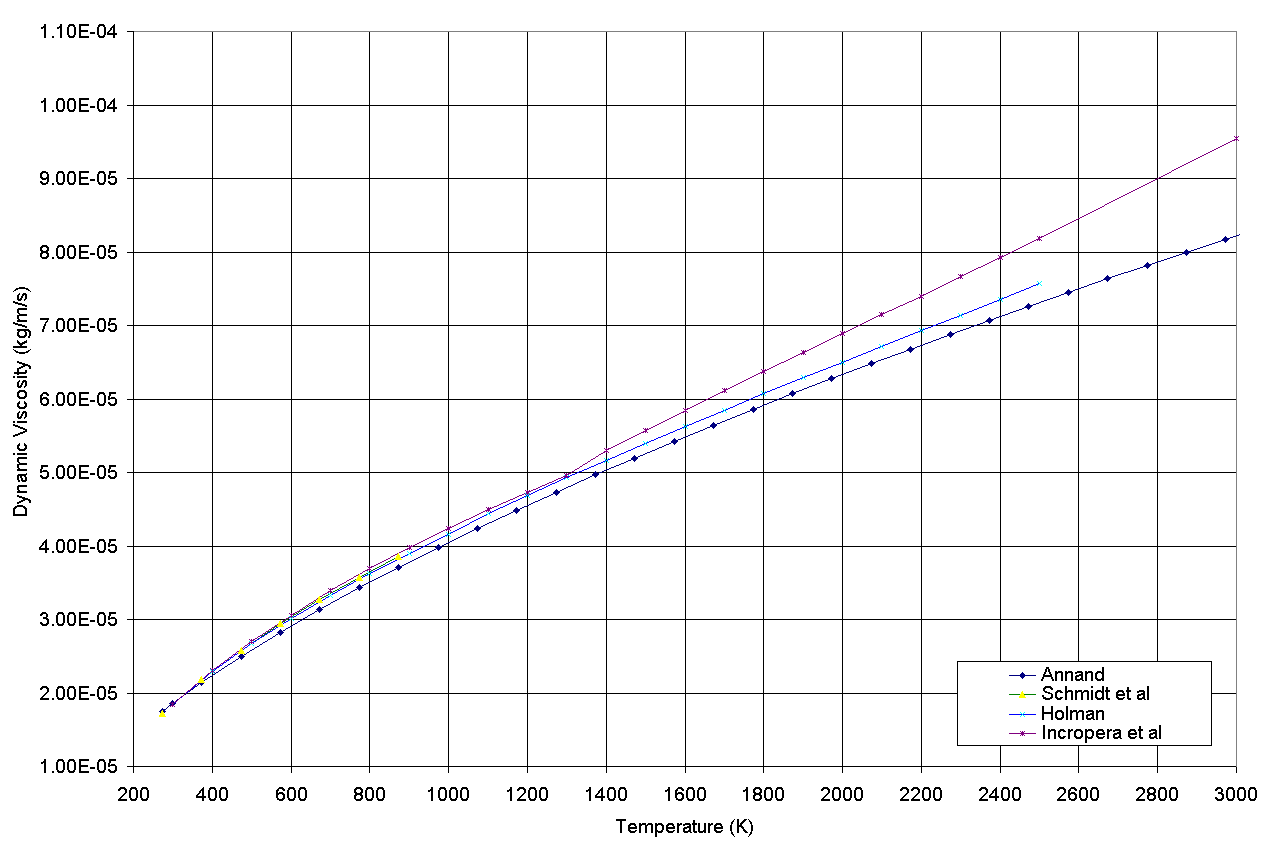
Index Tables Reference Viscopedia Data from. The viscosity of water decreases smoothly from the freezing point while the density remains essentially constant except for the small maximum in density near 4C. In cgs units the unit is called the centistoke. In cgs units the unit is called the centistoke. The kinematic viscosity is the dynamic viscosity divided by the density of the fluid. 16 To standardize comparisons among experiments and theoretical models. The kinematic viscosity is the dynamic viscosity divided by the density of the fluid. 4 Definition Dynamic viscosity Illustration of a planar Couette flow. The unit centiPoise = 0.001 Pa s and is is sometimes written mPa s. These viscosities are at 20☌ except for the blood and blood plasma which are at body temperature, 37☌, and for steam which is at 100☌. The Poise is used in the table because of its more common usage. More commonly used is the dyne sec/cm 2 which is called Poise. Viscosity has the SI units Pascal seconds (Pa s) which is called the Poiseuille. Both are stirred rapidly and kept.Viscosity of Liquids and Gases Viscosity of Liquids and Gases Liquids Glycerine and water are kept in separate containers. Kinematic viscosity is defined by the ratio of dynamic viscosity and fluid density. The kinematic viscosity equation can explain the relation between viscosity and density. 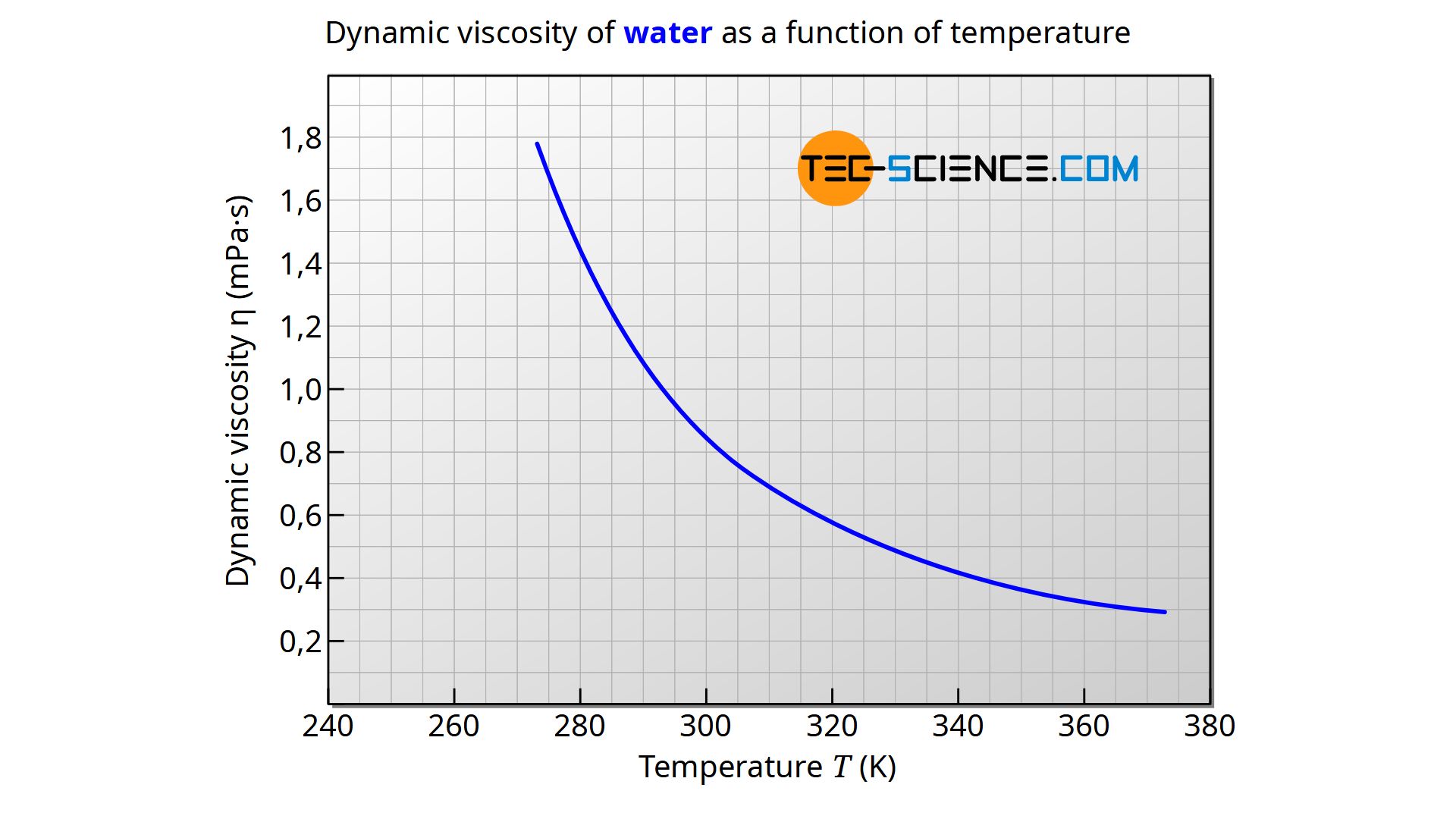
17, Volume fraction of water-vapour in flue gas r(water-vapour) : 23.72. In liquids, viscosity increases with increasing density. 16, Volume fraction of air in flue gas r(air) : 76.28. This, in turn, makes the fluid less viscous. When the temperature increases, the density of the fluid decreases. In other words, the rising temperature will make the molecules move apart, and the friction between the layers decreases. NOTE: Kinematic viscosity of air is about is times greater than the corresponding. 286 Joules per gram per degree Kelvin or 53.5 foot-pounds per pound per degree Rankine. This makes the molecules collide with each other, and hence the viscosity is decreased. In MKS units The dynamic viscosity are at standard condition. Temperature: 15 degrees Celsius or 59 degrees Farenheit Absolute Temperature: 288 degrees Kelvin or 519 degrees Rankine Viscosity: 1.73 time 10-5 Newton-second per square meter or 3.62 times 10-7 pound-second per square foot. 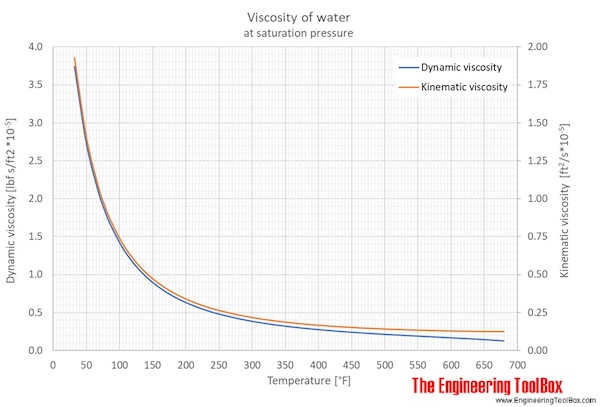
Welcome This video helps you to understand the. Then predict the viscosity of carbon dioxide at 100. Dynamic Viscosity & Kinematic Viscosity Formula Explained Fluid MechanicsHi Friends. On rising its temperature, the molecules within it start moving faster. Determine the constants a and b of Sutherland correlation for carbon dioxide at atmospheric pressure. First, let us freeze it, and now the molecules are attached. Both the viscosity and density are not directly related, but they are related in terms of temperature. Kinematic viscosity is defined as the ratio of dynamic viscosity and fluid density.The relation between viscosity and density can be explained by the kinematic viscosity equation.Both viscosity and density are not directly related, but they are related in terms of temperature.In other words, the crowding of molecules at the molecular level. Density tells how closely the molecules are packed together.Density is a fluid property to measure the distance between two particles in a fluid, i.e., it is the measure of mass per volume.Viscosity tells how difficult for the molecules to move relative to each other.Viscosity is a fluid property to measure the thickness of a fluid, i.e., it is a property that opposes the relative motion between the layers in a fluid which are moving at different velocities.These two tables show that viscosity decreases with decreasing density in liquids, whereas viscosity increases with decreasing density in gases. lates thermodynamic properties such as density and vapour pressure. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
